The septum is the wall separating the right side of the heart from the left side. The atrial septum divides the right and left atria and the ventricular septum divides the right and left ventricle. In a fetal heart, the foramen ovale (2 in the diagram), a small hole located in the atrial septum, allows blood to bypass the fetal lungs and travel directly to the rest of the body. This is because the fetus gets oxygen from the umbilical cord, not the fetal lungs. The foramen ovale usually closes within 48 hours of birth due to pressure changes in the infant’s chest. If the foramen ovale does not close, it is called a patent foramen ovale (PFO).
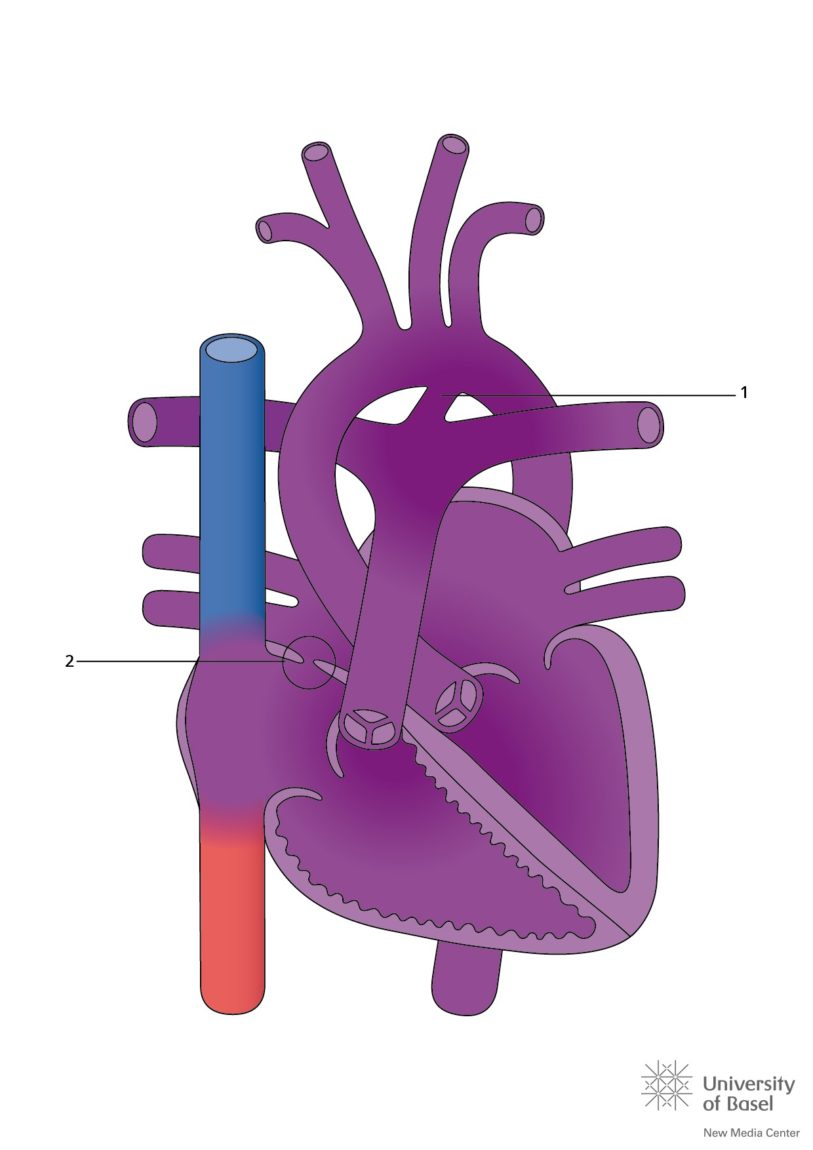
An atrial septal defect (ASD) occurs when part of the atrial septum does not form properly during development.
Repairs
Sometimes, depending on the type of ASD and PFO, the hole can be closed without open heart surgery. This is called a percutaneous repair and it is done in the cardiac catheterization lab. The procedure will typically take 1-2 hours to complete and may require the use of general anesthesia, meaning you will be asleep and won’t remember the procedure.
For a percutaneous repair, a catheter (a small tube) is placed into your groin and is threaded into your heart. The closure device that will plug the hole is folded into a special delivery catheter, which is threaded through the catheter to your heart, and through the defect, guided by fluoroscopy and echocardiography. When the catheter is in the right position, the closure device is slowly is pushed out of the catheter until the discs of the device sit on each side of the defect, like a sandwich.


